
| Sortilin (Soluble) ELISA Kit | |
| Sortilin (Soluble) Is A New Biomarker for Cardiovascular Diseases, Obesity & Cancer | |
Alternative name:
|
|
|
|
|
Sortilin deficiency improves the metabolic phenotype and reduces hepatic steatosis of mice subjected to diet-induced obesity |
|
BACKGROUND & AIMS: Sortilin traffics newly synthesized molecules from the trans-Golgi apparatus along secretory pathways to endosomes, lysosomes or to the cell surface. Sortilin trafficking of acid sphingomyelinase (aSMase) may regulate ceramide levels, a major modulator of insulin signalling. We therefore tested whether sortilin deficiency reduces hepatic and adipose tissue aSMase activity, improving insulin sensitivity in diet-induced obesity (DIO). METHODS:DIO in C57BL/6 (WT) and sortilin(-/-) mice was induced by high-fat diet feeding for 10weeks. RESULTS: Sortilin(-/-) mice gained less body weight and less visceral fat, despite similar food intake compared to WT type mice and had enhanced glucose uptake in insulin tolerance tests, which was further corroborated by enhanced hepatic pAkt expression. Sortilin deficiency led to attenuated hepatic steatosis, reduced expression of genes involved in lipogenesis, ceramide synthesis and inflammatory cytokine production and reduced activity of ceramide synthase 5/6 (CerS5/6). Sortilin(-/-) mice had reduced hepatic aSMase activity under both steady-state and DIO. Likewise, sortilin(-/-) hepatocytes displayed hypersensitivity to insulin, due to enhanced insulin receptor downstream signalling. In adipose tissue, sortilin(-/-) mice exhibited lower expression of inflammatory cytokines and lower expression and activity of CerS5/6. As in liver, adipose tissue displayed increased insulin signalling, accompanied by attenuated aSMase activity. CONCLUSIONS:Sortilin deficiency induces a beneficial metabolic phenotype in liver and adipose tissue upon DIO, mediated in part by reduced aSMase activity. |
|
Rabinowich L., et al. J Hepatol. 2015 Jan;62(1):175-81 |
|
Sortilin: the mechanistic link between genes, cholesterol, and coronary artery disease |
|
Hyperlipidemia is an important risk factor for cardiovascular disease globally, but there is still much mystery surrounding the topic of lipid regulation. Many studies have attempted to assess the underlying genetic basis of low-density lipoprotein (LDL) metabolism. Recently, multiple genome-wide association studies identified genes that strongly associate with plasma lipid concentration and cardiovascular disease. Compelling evidence linking the SORT1 gene to both LDL cholesterol (LDL-C) levels and the risk of coronary artery disease emerged from the data, prompting the search for the molecules and mechanisms responsible for this association. Three recent studies explored this relation through sortilin, the gene product of SORT1, and an intracellular trafficking molecule. Careful, hypothesis-driven experimental designs elucidated the potential mechanisms of sortilin's role in LDL-C metabolism. However, each study's conclusions differed in the details of SORT1's association to LDL-C and the subcellular mechanisms at work. Nevertheless, these 3 studies demonstrate how a complex disease such as hyperlipidemia can be evaluated from the scope of the genome down through the level of cellular regulation. Their findings serve as a platform for further study of LDL-C metabolism and hyperlipidemia while also providing lessons on how to better study other complex diseases. |
|
Thaker AM, Frishman WH.. Cardiol Rev. 2014 Mar-Apr;22(2):91-6. |
|
Sortilin and the risk of cardiovascular disease |
|
Plasma low-density lipoprotein cholesterol (LDL-C) levels are a key determinant of the risk of cardiovascular disease, which is why many studies have attempted to elucidate the pathways that regulate its metabolism. Novel latest-generation sequencing techniques have identified a strong association between the 1p13 locus and the risk of cardiovascular disease caused by changes in plasma LDL-C levels. As expected for a complex phenotype, the effects of variation in this locus are only moderate. Even so, knowledge of the association is of major importance, since it has unveiled a new metabolic pathway regulating plasma cholesterol levels. Crucial to this discovery was the work of three independent teams seeking to clarify the biological basis of this association, who succeeded in proving that SORT1, encoding sortilin, was the gene in the 1p13 locus involved in LDL metabolism. SORT1 was the first gene identified as determining plasma LDL levels to be mechanistically evaluated and, although the three teams used different, though appropriate, experimental methods, their results were in some ways contradictory. Here we review all the experiments that led to the identification of the new pathway connecting sortilin with plasma LDL levels and risk of myocardial infarction. The regulatory mechanism underlying this association remains unclear, but its discovery has paved the way for considering previously unsuspected therapeutic targets and approaches. |
|
Coutinho MF, et al. Rev Port Cardiol. 2013 Oct;32(10):793-9 |
|
Hepatic sortilin regulates both apolipoprotein B secretion and LDL catabolism |
|
Genome-wide association studies (GWAS) have identified a genetic variant at a locus on chromosome 1p13 that is associated with reduced risk of myocardial infarction, reduced plasma levels of LDL cholesterol (LDL-C), and markedly increased expression of the gene sortilin-1 (SORT1) in liver.Sortilin is a lysosomal sorting protein that binds ligands both in the Golgi apparatus and at the plasma membrane and traffics them to the lysosome. We previously reported that increased hepatic sortilin expression in mice reduced plasma LDL-C levels. Here we show that increased hepatic sortilinnot only reduced hepatic apolipoprotein B (APOB) secretion, but also increased LDL catabolism, and that both effects were dependent on intact lysosomal targeting. Loss-of-function studies demonstrated that sortilin serves as a bona fide receptor for LDL in vivo in mice. Our data are consistent with a model in which increased hepatic sortilin binds intracellular APOB-containing particles in the Golgi apparatus as well as extracellular LDL at the plasma membrane and traffics them to the lysosome for degradation. We thus provide functional evidence that genetically increased hepaticsortilin expression both reduces hepatic APOB secretion and increases LDL catabolism, providing dual mechanisms for the very strong association between increased hepatic sortilin expression and reduced plasma LDL-C levels in humans. |
|
Strong A et al. J Clin Invest. 2012 Aug 1;122(8):2807-16. doi: 10.1172/JCI63563. Epub 2012 Jul 2. |
|
Activation of ER stress and mTORC1 suppresses hepatic sortilin-1 levels in obese mice |
|
| Recent GWAS have identified SNPs at a human chromosom1 locus associated with coronary artery disease risk and LDL cholesterol levels. The SNPs are also associated with altered expression of hepatic sortilin-1 (SORT1), which encodes a protein thought to be involved in apoB trafficking and degradation. Here, we investigated the regulation of Sort1 expression in mouse models of obesity. Sort1 expression was markedly repressed in both genetic (ob/ob) and high-fat diet models of obesity; restoration of hepatic sortilin-1 levels resulted in reduced triglyceride and apoB secretion. Mouse models of obesity also exhibit increased hepatic activity of mammalian target of rapamycin complex 1 (mTORC1) and ER stress, and we found that administration of the mTOR inhibitor rapamycin to ob/ob mice reduced ER stress and increased hepatic sortilin-1 levels. Conversely, genetically increased hepatic mTORC1 activity was associated with repressed Sort1 and increased apoB secretion. Treating WT mice with the ER stressor tunicamycin led to marked repression of hepatic sortilin-1 expression, while administration of the chemical chaperone PBA to ob/ob mice led to amelioration of ER stress, increased sortilin-1 expression, and reduced apoB and triglyceride secretion. Moreover, the ER stress target Atf3 acted at the SORT1 promoter region as a transcriptional repressor, whereas knockdown of Atf3 mRNA in ob/ob mice led to increased hepatic sortilin-1 levels and decreased apoB and triglyceride secretion. Thus, in mouse models of obesity, induction of mTORC1 and ER stress led to repression of hepatic Sort1 and increased VLDL secretion via Atf3. This pathway may contribute to dyslipidemia in metabolic disease. | |
| Ai D, et al. J Clin Invest. 2012 May 1;122(5):1677-87. doi: 10.1172/JCI61248. Epub 2012 Apr 2. | |
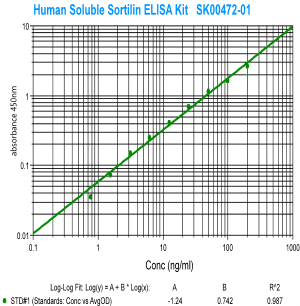 |
Human Sortilin ELISA Code No.: SK00472-01 Size: 96 T Price: $490.00 USD Standard Range:1.56-200 ng/ml Sensitivity:300 pg/ml Sample Type: cell culture, tissue homogenates Sample requres: 100uL per well IntraCV:4-6% InterCV: 8-12% Protocol: PDF |
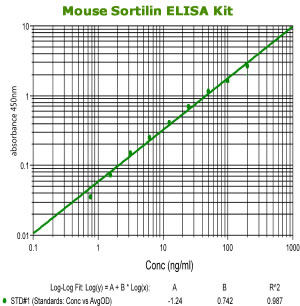 |
Mouse Sortilin ELISA Code No.: SK00472-03 Size: 96 T Price: $490.00 USD Standard Range:1.56-200 ng/ml Sensitivity:300 pg/ml Sample Type: cell culture, tissue homogenates Sample requres: 100uL per well IntraCV:4-6% InterCV: 8-12% Protocol: PDF |
|
|
| Name | Code No. |
Size |
Price ($) |
| Soluble Sortilin (Human) ELISA Kit | 96 T |
490.00 |
|
| Soluble Sortilin (Mouse) ELISA Kit | 96 T |
460.00 |
| References |
|